Blog
Orbion Team
How to Remove Endotoxin from Recombinant Protein

Your protein is pure. SDS-PAGE shows a single band. Activity is fine. Then you add it to your cell-based assay and everything lights up—NF-κB activation, cytokine storm, cells dying. You don't have a toxic protein. You have an endotoxin problem.
Endotoxin (lipopolysaccharide, LPS) is the most common contaminant that ruins cell-based experiments with E. coli-expressed proteins. It's invisible on gels, doesn't affect most biochemical assays, and is shockingly hard to remove. Here's what actually works.
Key Takeaways
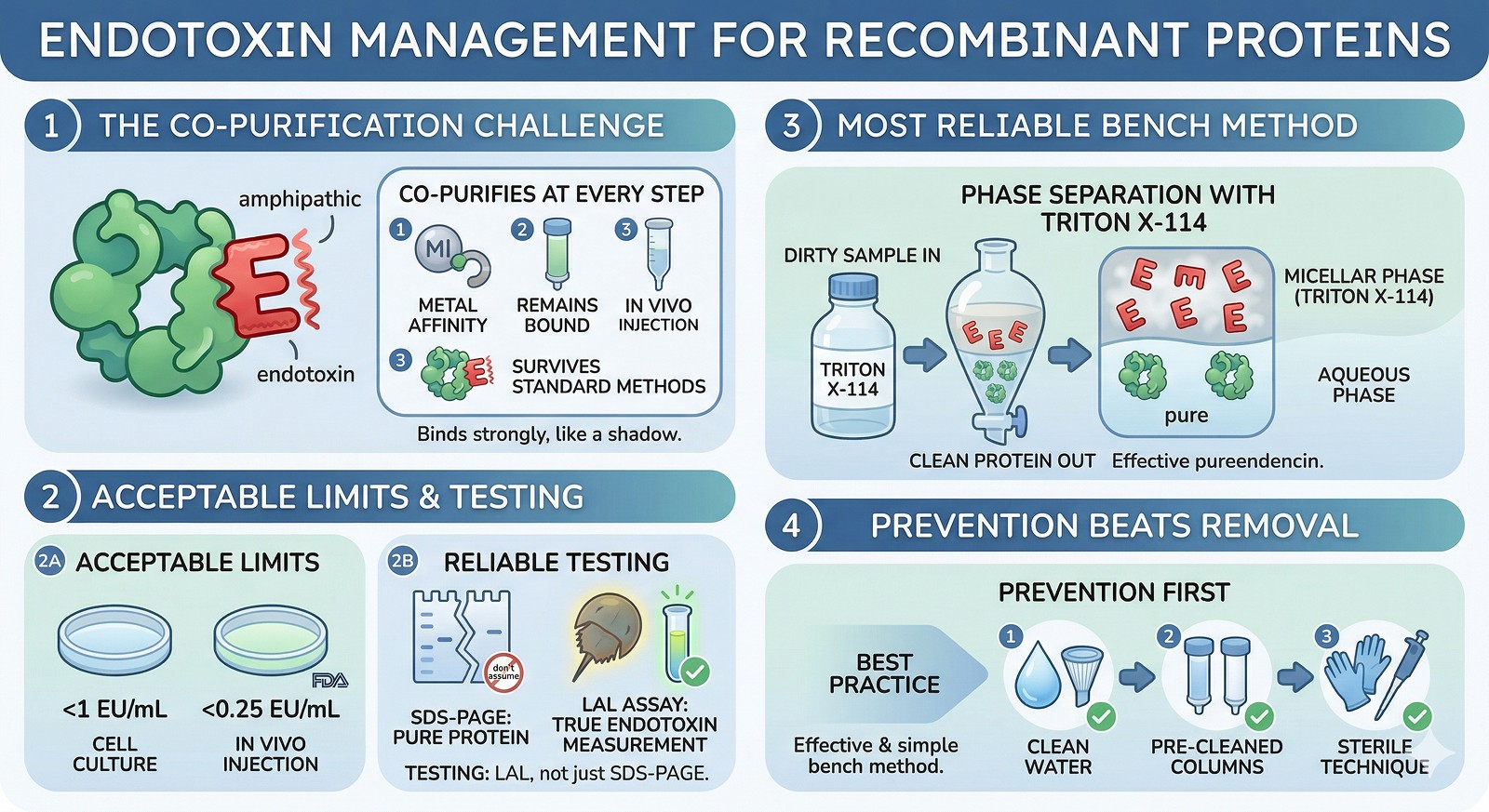
Endotoxin co-purifies with proteins at every step—it's amphipathic, binds metal affinity resins, and survives standard purification
Acceptable limits: <1 EU/mL for cell culture, <0.25 EU/mL for in vivo injection (FDA guideline: 5 EU/kg/hr)
The LAL assay is your only reliable test—don't assume your protein is clean because it looks pure on SDS-PAGE
Phase separation with Triton X-114 is the most reliable bench-scale method—cheap, effective, works for most proteins
Prevention beats removal: use endotoxin-free water, pre-cleaned columns, and handle everything with endotoxin-free technique from the start
Why Endotoxin Is So Problematic
LPS is a lipid-anchored glycan from the outer membrane of gram-negative bacteria. Key properties:
Amphipathic: hydrophobic lipid A core + hydrophilic polysaccharide chain → co-purifies with both hydrophobic and hydrophilic proteins
Thermostable: survives autoclaving (121°C, 30 min only reduces ~10-fold)
Binds metal resins: LPS interacts with Ni-NTA and Co-NTA columns, co-eluting with His-tagged proteins
Extremely potent: TLR4 responds to picogram quantities; even "trace" contamination can activate immune cells

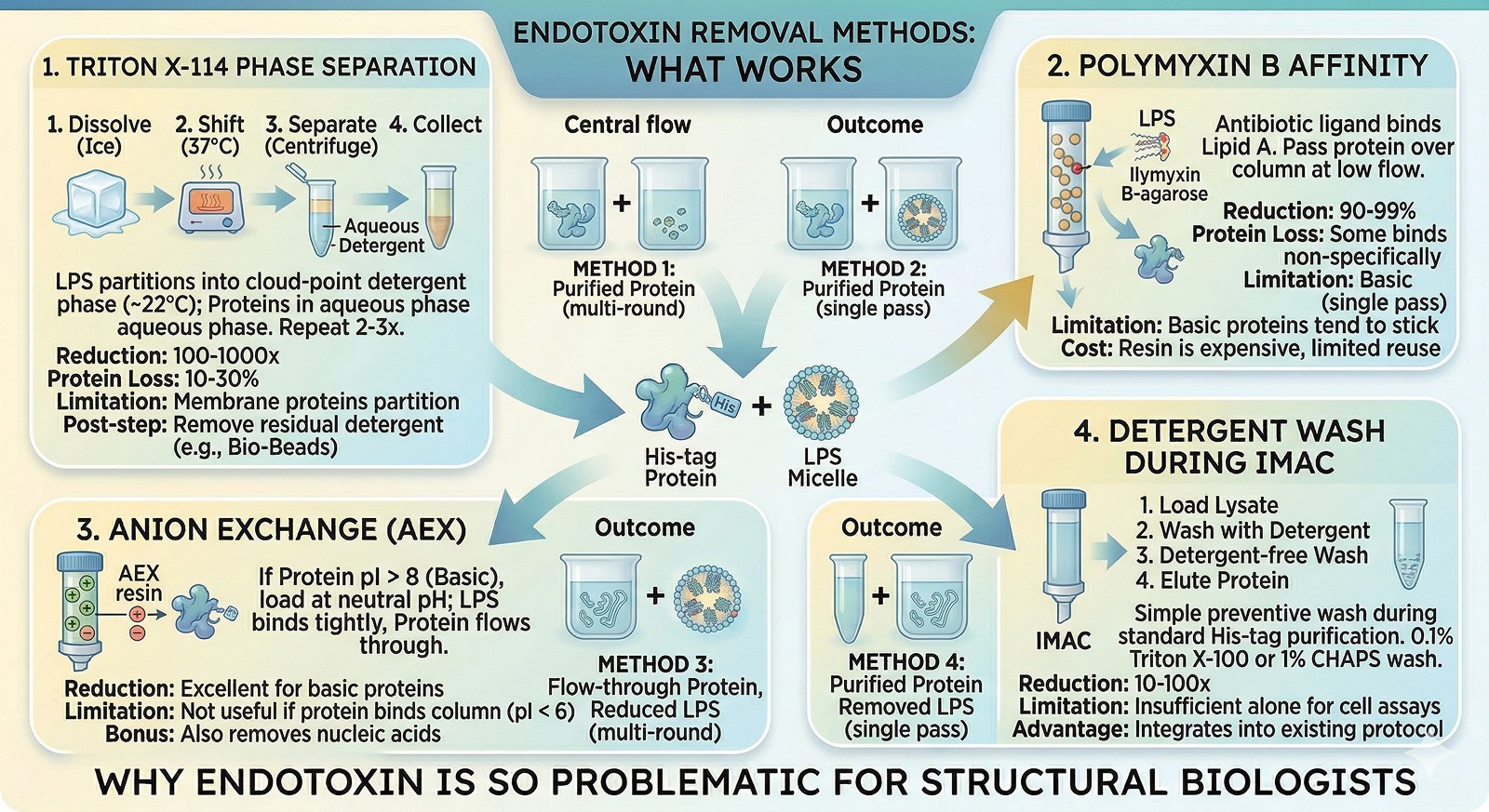
Removal Methods: What Works
Method 1: Triton X-114 Phase Separation (Best General Method)
Triton X-114 forms micelles above its cloud point (~22°C). LPS partitions into the detergent phase; most soluble proteins stay in the aqueous phase.
Protocol:
Add Triton X-114 to 1% (v/v) to your protein solution
Incubate on ice for 5 min (detergent dissolves)
Shift to 37°C for 10 min (phase separation occurs—solution goes cloudy)
Centrifuge at 20,000 × g for 10 min at 25°C
Carefully collect the upper aqueous phase (your protein)
Repeat 2–3 times for maximum removal
Effectiveness: 100–1000-fold reduction per round; 2–3 rounds typically reach <1 EU/mL
Protein loss: 10–30% per round (some protein partitions into detergent phase)
Limitation: Membrane proteins and very hydrophobic proteins will partition with the LPS
Residual detergent: Remove remaining Triton X-114 with Bio-Beads SM-2 or extensive dialysis
Method 2: Polymyxin B Affinity Chromatography
Polymyxin B is a cyclic peptide antibiotic that binds the lipid A moiety of LPS with high affinity.
Format: Polymyxin B-agarose columns (commercial, e.g., Pierce)
Protocol: Pass protein over column at slow flow rate (0.5 mL/min); collect flow-through
Effectiveness: 90–99% removal in a single pass
Limitation: Some proteins bind polymyxin B non-specifically → protein loss. Positively charged proteins are particularly prone to sticking
Cost: Resin is expensive and has limited reuse cycles
Method 3: Anion Exchange Chromatography
LPS is highly negatively charged. Anion exchange (Q Sepharose, DEAE) binds LPS tightly.
Strategy: If your protein is positively charged (pI > 8), load at neutral pH—protein flows through, LPS binds
Effectiveness: Excellent for basic proteins; less useful if your protein also binds the column
Bonus: Also removes nucleic acid contamination
Limitation: If your protein is acidic (pI < 6), both protein and LPS bind → harder to separate
Method 4: Detergent Washing During IMAC
The simplest preventive measure during His-tag purification.
Protocol: After loading and before elution, wash with 20 CV of buffer containing 0.1% Triton X-100 or 1% CHAPS, followed by 10 CV of detergent-free buffer
Effectiveness: 10–100-fold reduction (not sufficient alone for cell assays, but a good first step)
Advantage: No extra purification step; integrates into existing protocol
Method Comparison
Method | LPS Removal | Protein Loss | Best For | Cost |
|---|---|---|---|---|
Triton X-114 phase separation | 100–1000× per round | 10–30% per round | Most soluble proteins | Low |
Polymyxin B agarose | 90–99% single pass | Variable (5–50%) | When you need speed | High |
Anion exchange | >99% for basic proteins | Low if pI > 8 | Basic proteins (pI > 8) | Medium |
IMAC detergent wash | 10–100× | Minimal | Prevention during IMAC | Low |
Activated carbon | Variable | High (30–60%) | Last resort | Low |
Measuring Endotoxin: The LAL Assay
The Limulus Amebocyte Lysate (LAL) assay is the gold standard.
Sensitivity: Down to 0.005 EU/mL (kinetic turbidimetric method)
Formats: Gel-clot (qualitative), chromogenic (quantitative), kinetic turbidimetric (most sensitive)
Critical controls: Always run a positive product control (PPC)—your protein may inhibit or enhance the LAL reaction
Common pitfall: High protein concentrations can mask endotoxin → dilute your sample and re-test
Target values:
Application | Maximum Acceptable Endotoxin |
|---|---|
Cell culture (immune cells) | <0.1 EU/mL |
Cell culture (non-immune) | <1 EU/mL |
In vivo injection (mice) | <0.5 EU/dose |
Parenteral drugs (FDA) | <5 EU/kg/hr |

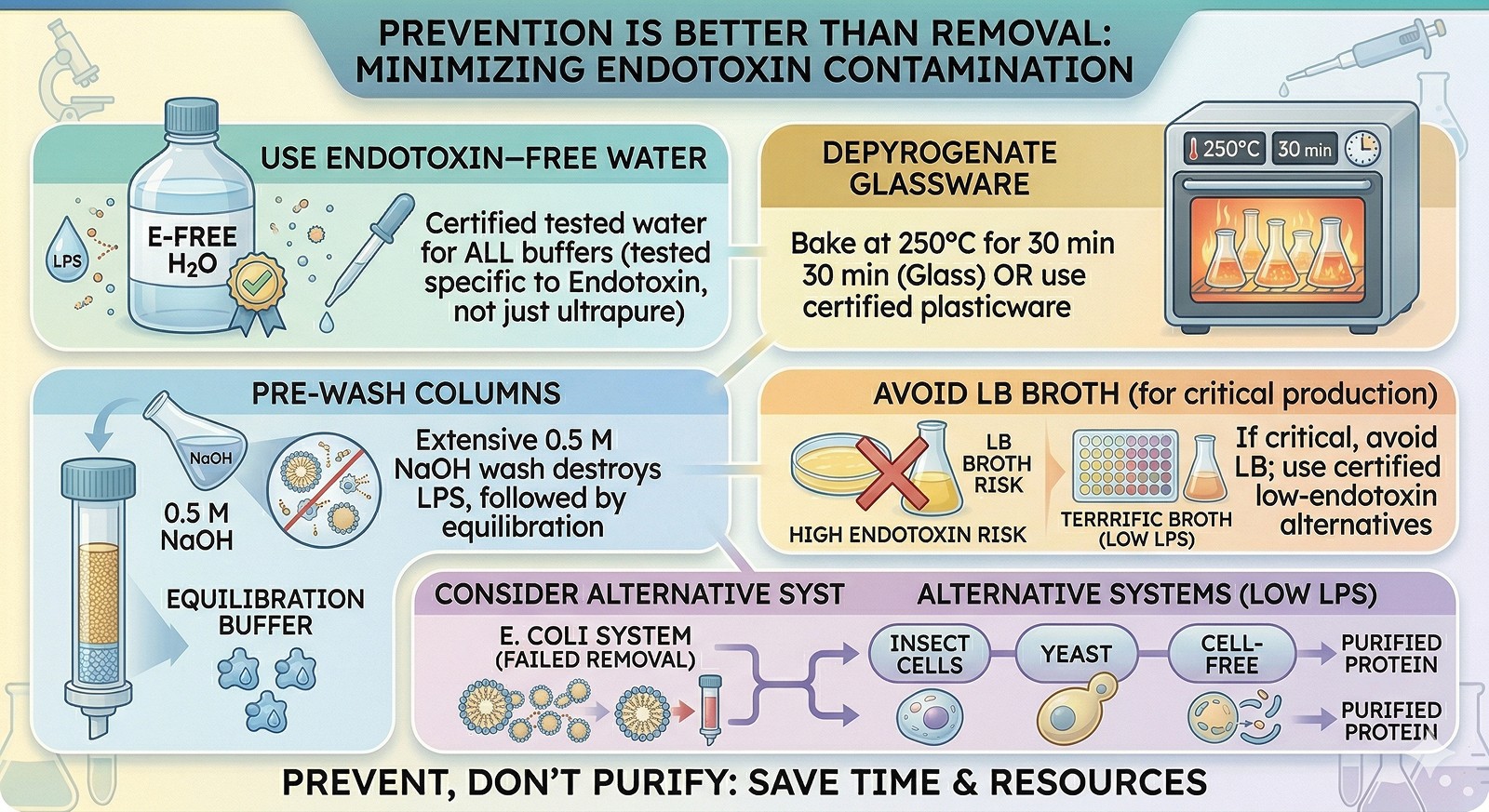
Prevention Is Better Than Removal
Use endotoxin-free water for all buffer preparation (not just "ultrapure"—specifically tested for endotoxin)
Depyrogenate glassware by baking at 250°C for 30 min (or use certified endotoxin-free plasticware)
Pre-wash columns extensively with 0.5 M NaOH (destroys LPS), then equilibrate
Avoid LB broth for production cultures if endotoxin is critical—or accept that you'll need aggressive removal
Consider alternative expression systems (insect cells, yeast, cell-free) if endotoxin removal consistently fails
The Bottom Line
Situation | Recommended Approach |
|---|---|
His-tagged soluble protein for cell assays | IMAC detergent wash + 2–3 rounds Triton X-114 |
Basic protein (pI > 8) for in vivo use | Anion exchange (Q column) at pH 7 |
High-value protein, can't afford loss | Polymyxin B column (single pass, gentle) |
Membrane protein | Switch expression system (insect cells); phase separation won't work |
Persistent endotoxin despite removal | Check buffers, water, and plasticware; re-purify from scratch with clean materials |
The one rule: Test with a LAL assay before using any E. coli-produced protein in cell-based or in vivo experiments. Assumptions about purity will eventually burn you.

Planning Endotoxin-Free Workflows
Orbion's Bench module generates detailed purification protocols that include endotoxin removal steps when the downstream application requires it. By specifying your target application upfront—cell assay, in vivo study, or structural biology—the protocol adapts to include the appropriate removal methods, wash steps, and quality control checkpoints.
References
Magalhães PO, et al. (2007). Methods of endotoxin removal from biological preparations: a review. Journal of Pharmacy and Pharmaceutical Sciences, 10(3):388-404. Link
Aida Y, Pabst MJ. (1990). Removal of endotoxin from protein solutions by phase separation using Triton X-114. Journal of Immunological Methods, 132(2):191-195. Link
Petsch D, Anspach FB. (2000). Endotoxin removal from protein solutions. Journal of Biotechnology, 76(2-3):97-119. Link
