Blog
Orbion Team
Why Does My Protein Aggregate at 4°C?

You purified your protein at room temperature. It looked great—single peak on SEC, clear solution, good activity. You put it in the fridge overnight. The next morning, it's cloudy. Or there's a pellet at the bottom of the tube. Or the SEC profile now shows a void volume peak that wasn't there yesterday.
Cold-induced aggregation is surprisingly common, counterintuitive, and entirely preventable once you understand why it happens.
Key Takeaways
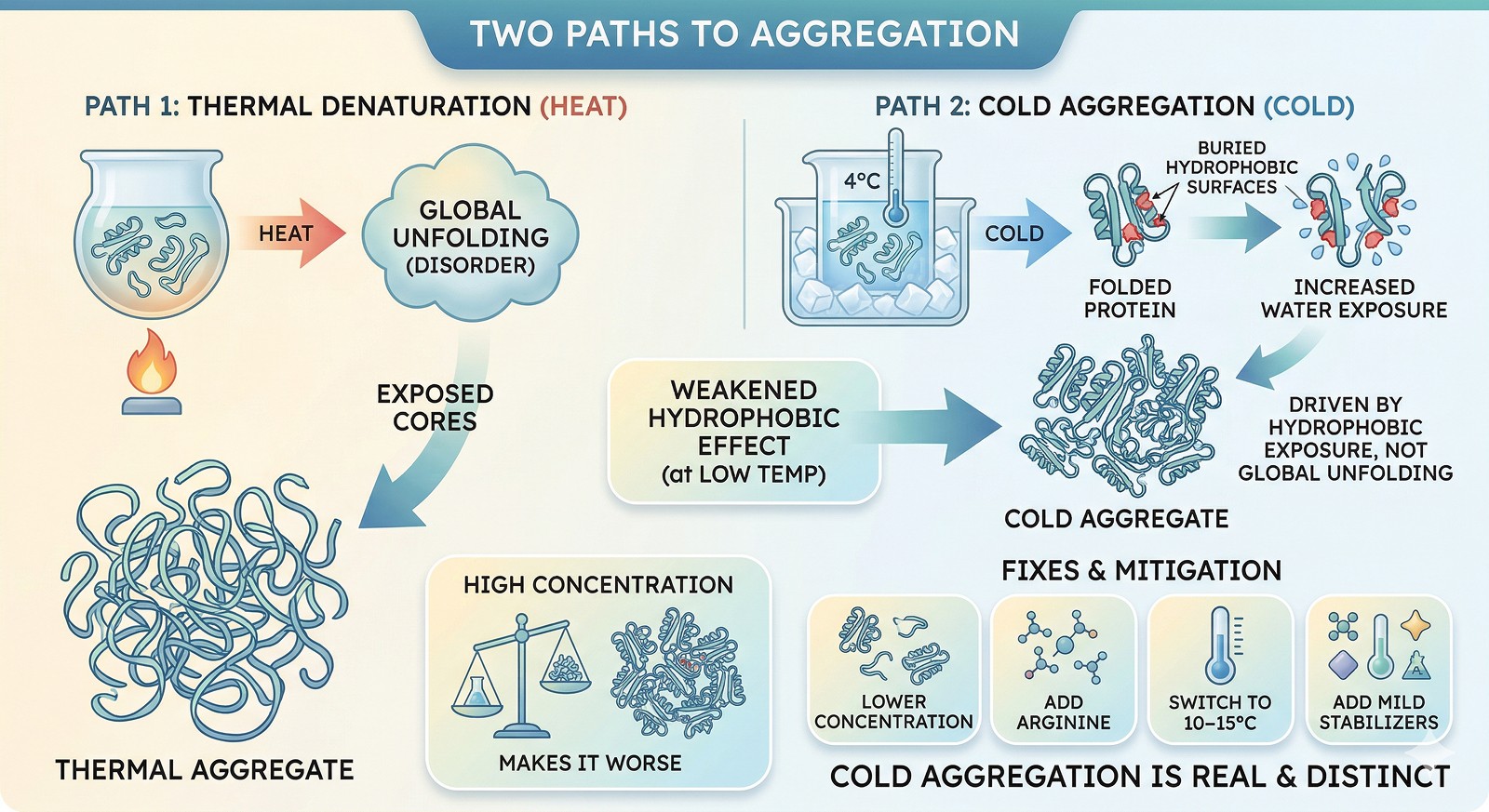
Cold aggregation is real and distinct from thermal aggregation—it's driven by hydrophobic exposure, not unfolding
The hydrophobic effect weakens at low temperature—buried hydrophobic surfaces become more exposed at 4°C
Proteins near their cold denaturation temperature are vulnerable—this is a thermodynamic reality, not a storage error
High concentration makes it worse—cold aggregation is concentration-dependent because it requires intermolecular contacts
Fixes exist: lower the concentration, add arginine, switch to 10–15°C storage, or add mild stabilizers
Why Proteins Aggregate in the Cold
The Hydrophobic Effect Has a Temperature Dependence
The hydrophobic effect—the force that buries nonpolar amino acids in the protein core—has a temperature optimum. Below that optimum, hydrophobic interactions weaken.
At 4°C compared to 25°C:
Hydrophobic core packing is slightly loosened
Partially exposed hydrophobic patches become more accessible to solvent
These exposed patches drive intermolecular association → aggregation
This is the mirror image of heat denaturation. At high temperature, the protein unfolds. At low temperature, the protein partially unfolds in a different way—and the exposed surfaces are "sticky."
Cold Denaturation Is Thermodynamically Real
Every protein has both a high-temperature unfolding transition (Tm) and a low-temperature unfolding transition (Tc, cold denaturation temperature). For most proteins, Tc is well below 0°C and doesn't matter. But for marginally stable proteins:
Tc can be 0–10°C
At 4°C, these proteins are partially cold-denatured → aggregation-prone
This is especially common for engineered proteins, truncation constructs, and mutants with reduced stability
Common Scenarios and Fixes
Scenario 1: Concentrated Protein Precipitates Overnight at 4°C
What's happening: High concentration + cold temperature exceeds the colloidal stability limit. Weak hydrophobic interactions between partially exposed surfaces drive reversible association that becomes irreversible precipitation over hours.
Fixes:
Dilute to <1 mg/mL before storing at 4°C
Add 150–300 mM arginine (shields hydrophobic patches without destabilizing fold)
Store at 10–15°C instead of 4°C (if short-term)
Flash-freeze in liquid nitrogen with 10% glycerol for long-term storage
Scenario 2: SEC Shows Void Volume Peak After Cold Storage
What's happening: Soluble aggregates formed at 4°C. These are large enough to elute in the void volume but may not be visible as turbidity.
Diagnosis: Compare SEC profiles before and after cold storage. If void volume peak appears or grows, cold aggregation is occurring.
Fixes:
Add 200 mM arginine + 200 mM glutamate (Arg/Glu system—best general aggregation suppressor)
Switch to 5% glycerol or 5% sucrose as cryoprotectant
Filter (0.22 µm) before SEC to remove large aggregates, but this doesn't fix the underlying problem
Scenario 3: Protein Is Fine Fresh But Aggregates After Freeze-Thaw
This is freeze-thaw damage, not cold aggregation per se—but the result is the same. See the companion article on protein activity loss after freezing.
Scenario 4: Protein Aggregates Only at High Concentration
What's happening: Self-association is concentration-dependent. At low concentration (<0.5 mg/mL), the protein is fine. Above a threshold (often 2–10 mg/mL), intermolecular contacts overcome the entropic cost of association.
The cold makes it worse: The critical concentration threshold is lower at 4°C than at 25°C because hydrophobic interactions that drive aggregation are relatively stronger compared to the weakened intramolecular hydrophobic effect.
Fixes:
Determine your protein's maximum stable concentration at 4°C empirically (SEC at increasing concentrations)
Add 200–500 mM NaCl (ionic strength screens electrostatic attraction)
Add 5–10% glycerol (preferential exclusion stabilizes compact state)
The Stabilizer Toolkit
Additive | Concentration | Mechanism | Best For |
|---|---|---|---|
Arginine | 150–300 mM | Shields hydrophobic patches | General anti-aggregation |
Glycerol | 5–10% (v/v) | Preferential exclusion; stabilizes compact fold | Broad stabilization |
Sucrose/Trehalose | 5–10% (w/v) | Same as glycerol; also protects during freeze-thaw | Storage stability |
NaCl | 150–500 mM | Screens electrostatic interactions | Charge-driven aggregation |
Arg + Glu | 200 mM each | Synergistic; suppresses both hydrophobic and electrostatic aggregation | Stubborn cases |
Mild detergent | 0.01% Tween-20 | Prevents surface adsorption; blocks hydrophobic patches | Dilute protein solutions |
When Cold Storage Actually Works
Not all proteins aggregate at 4°C. Cold storage is fine when:
The protein has a high Tm (>60°C) and therefore a very low Tc
The protein is at moderate concentration (<2 mg/mL) in a good buffer
The protein has low surface hydrophobicity (few exposed nonpolar patches)
Storage is short-term (<1 week)
For long-term storage: Flash-freeze aliquots in liquid nitrogen with cryoprotectant. This avoids both cold aggregation and the slow degradation that happens at 4°C over weeks.
The Bottom Line
Observation | Likely Cause | Fix |
|---|---|---|
Precipitation at 4°C overnight | Cold-induced hydrophobic exposure | Dilute; add arginine; store at 10–15°C |
Void volume peak on SEC after storage | Soluble aggregate formation | Add Arg/Glu; add glycerol; reduce concentration |
Aggregation only at high concentration | Concentration-dependent self-association | Find maximum stable concentration; add stabilizers |
Aggregation after freeze-thaw | Ice crystal damage / freeze-concentration | Flash-freeze aliquots with cryoprotectant |
Fine at 25°C, aggregates at 4°C | Marginal stability near cold denaturation | Test storage at 10°C; screen stabilizers |
The one rule: If your protein aggregates at 4°C, don't just accept it and work with cloudy samples. Optimize your buffer, adjust the concentration, and test a few stabilizers—most cold aggregation problems are solvable in a day.
Predicting Aggregation Risk
Orbion's AstraUNFOLD predicts amyloidogenicity and aggregation-prone regions in your protein sequence, while AstraDTM and AstraDDG can evaluate how mutations affect stability—including the margin above cold denaturation. Knowing your protein's stability profile before purification lets you design storage conditions proactively rather than discovering aggregation problems the morning after.
References
Privalov PL. (1990). Cold denaturation of proteins. Critical Reviews in Biochemistry and Molecular Biology, 25(4):281-305. Link
Tsumoto K, et al. (2004). Role of arginine in protein refolding, solubilization, and purification. Biotechnology Progress, 20(5):1301-1308. Link
Arakawa T, et al. (2007). Suppression of protein interactions by arginine: a proposed mechanism of the arginine effects. Biophysical Chemistry, 127(1-2):1-8. Link
