Blog
Orbion Team
Why Does My Protein Lose Activity After Freezing

You purified 10 mg of enzyme last month. Activity was perfect: kcat = 45 s⁻¹. You aliquoted, flash-froze in liquid nitrogen, and stored at –80°C. Today you thawed an aliquot. kcat = 8 s⁻¹. You thawed another. kcat = 12 s⁻¹. A third: 6 s⁻¹. Three aliquots from the same prep, all with different (and terrible) activity. Your freezer isn't preserving your protein—it's destroying it.
Freeze-thaw damage is silent, variable, and cumulative. Understanding why it happens lets you prevent it.
Key Takeaways
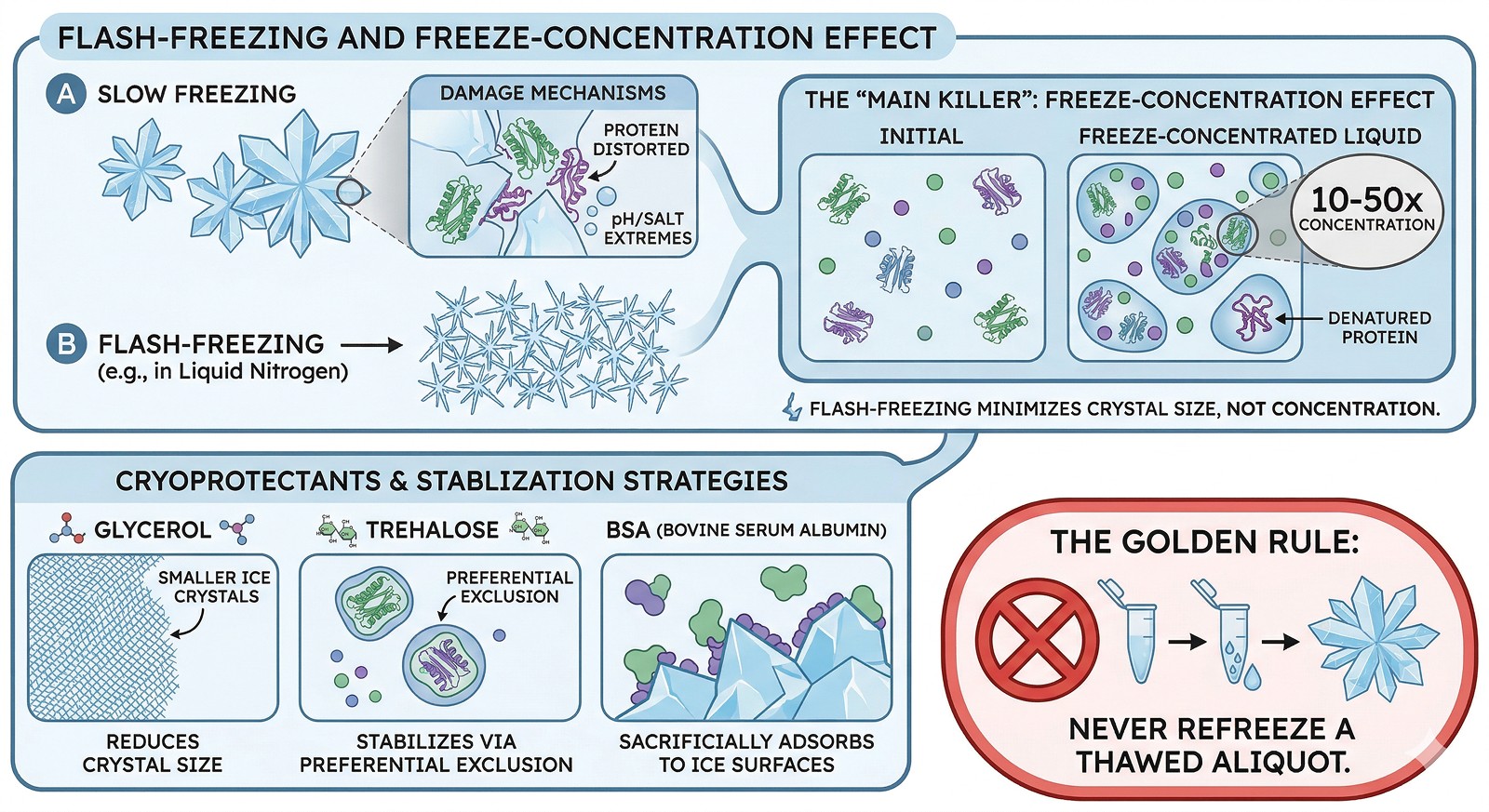
Ice crystal formation damages proteins mechanically by concentrating them at grain boundaries and creating local pH/salt extremes
The freeze-concentration effect is the main killer: as water freezes, solutes concentrate 10–50x in the remaining liquid, creating denaturing conditions
Flash-freezing helps but doesn't eliminate damage—small ice crystals are less damaging than large ones, but the concentration effect still occurs
Cryoprotectants work by different mechanisms: glycerol reduces ice crystal size, trehalose stabilizes through preferential exclusion, and BSA sacrificially adsorbs to ice surfaces
The single most important rule: never refreeze a thawed aliquot
What Happens During Freezing
The Freeze-Concentration Effect
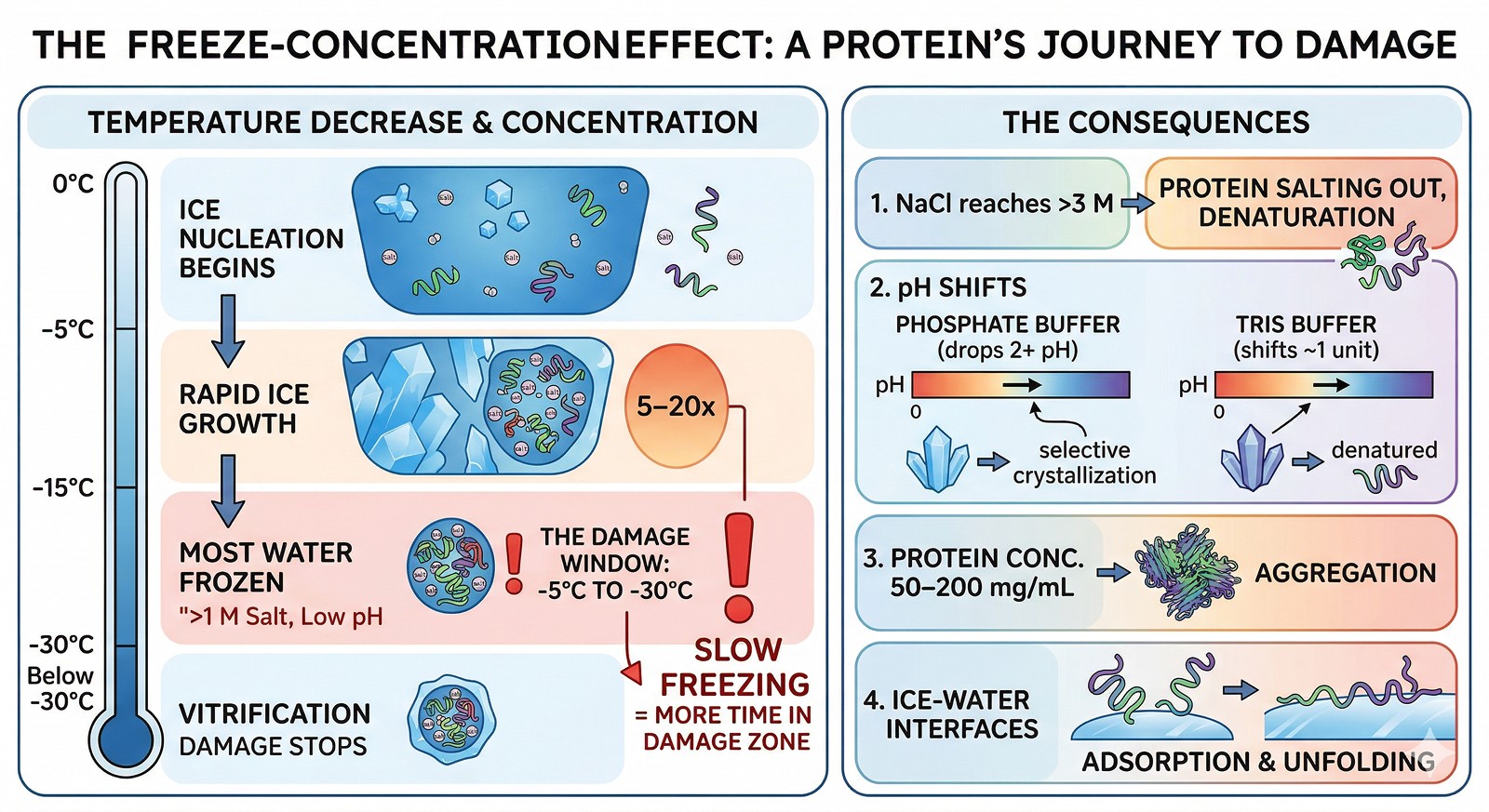
When a protein solution freezes, pure water crystallizes first. Everything else—salt, buffer, protein—concentrates in the remaining liquid phase (Chang et al., 2009).
As Temperature Drops | What Happens |
|---|---|
0°C to –5°C | Ice nucleation begins; solutes start concentrating |
–5°C to –15°C | Rapid ice growth; solute concentration increases 5–20x |
–15°C to –30°C | Most water is frozen; remaining liquid is highly concentrated (>1 M salt, low pH) |
Below –30°C | Remaining liquid vitrifies (glass transition); damage stops |
The damage window is –5°C to –30°C. Your protein spends time in this range during both freezing AND thawing. Slow freezing = more time in the damage zone.
What the Concentrated Environment Does
In the freeze-concentrated liquid:
NaCl can reach >3 M → protein salting out, denaturation
Buffer pH shifts: phosphate buffer can drop by 2+ pH units as dibasic phosphate crystallizes selectively (Pikal-Cleland et al., 2000). Tris shifts ~1 unit.
Protein concentration reaches 50–200 mg/mL → aggregation, non-native interactions
Ice-water interfaces provide surfaces for protein adsorption and unfolding
The Fix: Cryoprotection
The Essential Additives
Cryoprotectant | Concentration | Mechanism | Best For |
|---|---|---|---|
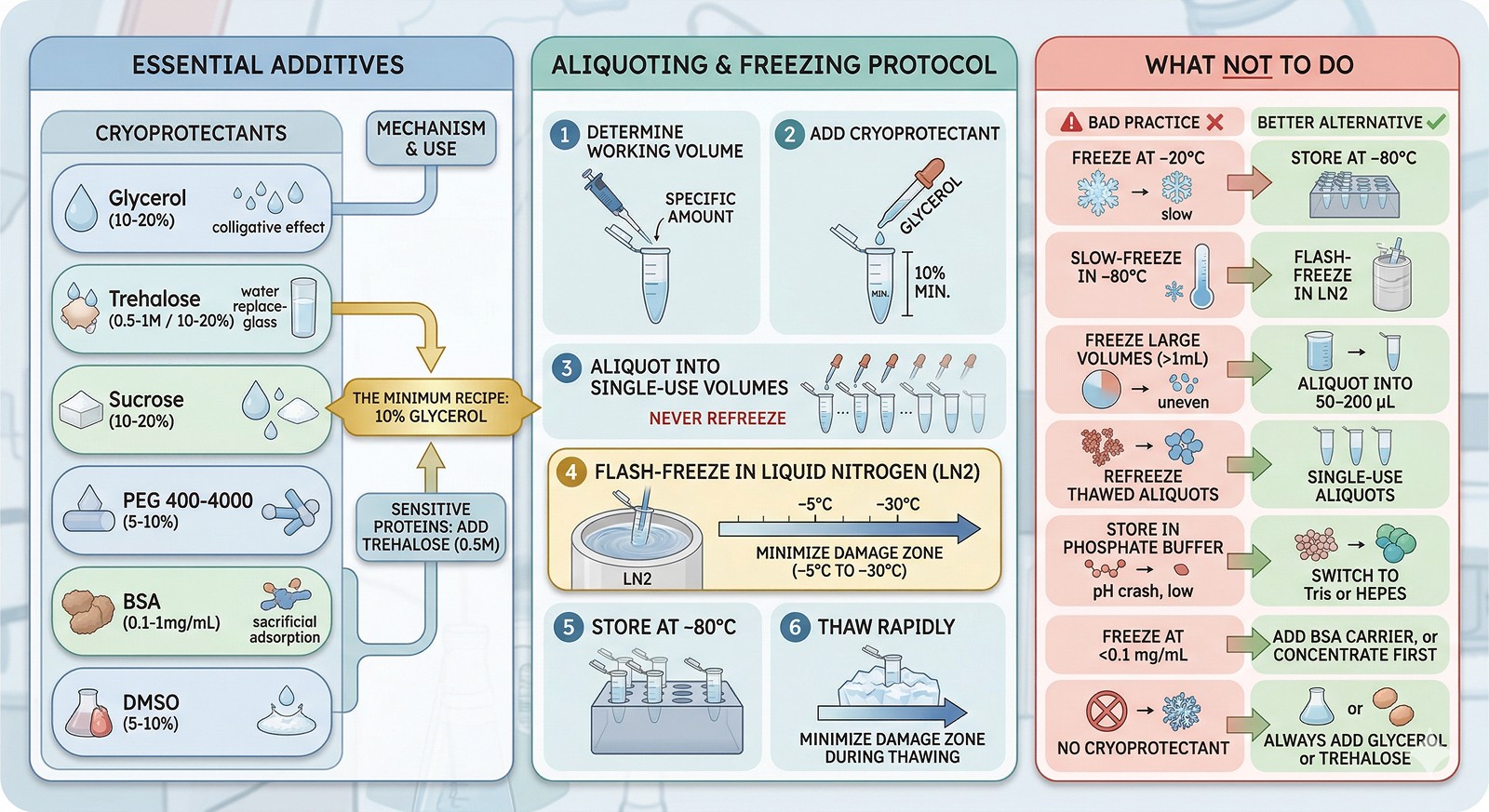
Glycerol | 10–20% | Reduces ice crystal size; colligative freezing point depression | General purpose; cheap |
Trehalose | 0.5–1 M (or 10–20%) | Preferential exclusion; vitrification; replaces water shell | Sensitive enzymes; lyophilization |
Sucrose | 10–20% | Same as trehalose | General purpose |
BSA | 0.1–1 mg/mL | Sacrificial protein; adsorbs to ice surfaces instead of your protein | Dilute protein solutions |
PEG 400–4000 | 5–10% | Reduces ice crystal growth | Some membrane proteins |
DMSO | 5–10% | Penetrating cryoprotectant; prevents intracellular ice | Cell-based applications (not ideal for purified protein) |
The minimum recipe: 10% glycerol. This alone prevents most freeze-thaw damage for most proteins. Add trehalose (0.5 M) for especially sensitive proteins.
The Aliquoting Protocol
Determine working volume (how much do you use per experiment?)
Add cryoprotectant before aliquoting (10% glycerol minimum)
Aliquot into single-use volumes (never refreeze)
Flash-freeze in liquid nitrogen (minimizes time in the –5°C to –30°C damage zone)
Store at –80°C (below the glass transition)
Thaw rapidly on ice or at room temperature (minimize time in the damage zone during thawing)
What NOT to Do
Bad Practice | Why It's Bad | Better Alternative |
|---|---|---|
Freeze at –20°C | Temperature is IN the damage zone; ice recrystallizes constantly | Always use –80°C |
Slow-freeze in a –80°C freezer | Hours in the damage zone | Flash-freeze in LN₂, then transfer to –80°C |
Freeze large volumes (>1 mL) | Slow freezing, uneven cryoprotection | Aliquot into 50–200 µL |
Refreeze thawed aliquots | Each cycle damages more protein | Single-use aliquots |
Store in phosphate buffer | pH crash during freezing (up to –2 units) | Switch to Tris or HEPES |
Freeze at <0.1 mg/mL | More surface adsorption damage at low concentration | Add BSA carrier, or concentrate first |
No cryoprotectant | Maximum ice crystal damage | Always add glycerol or trehalose |
Special Cases
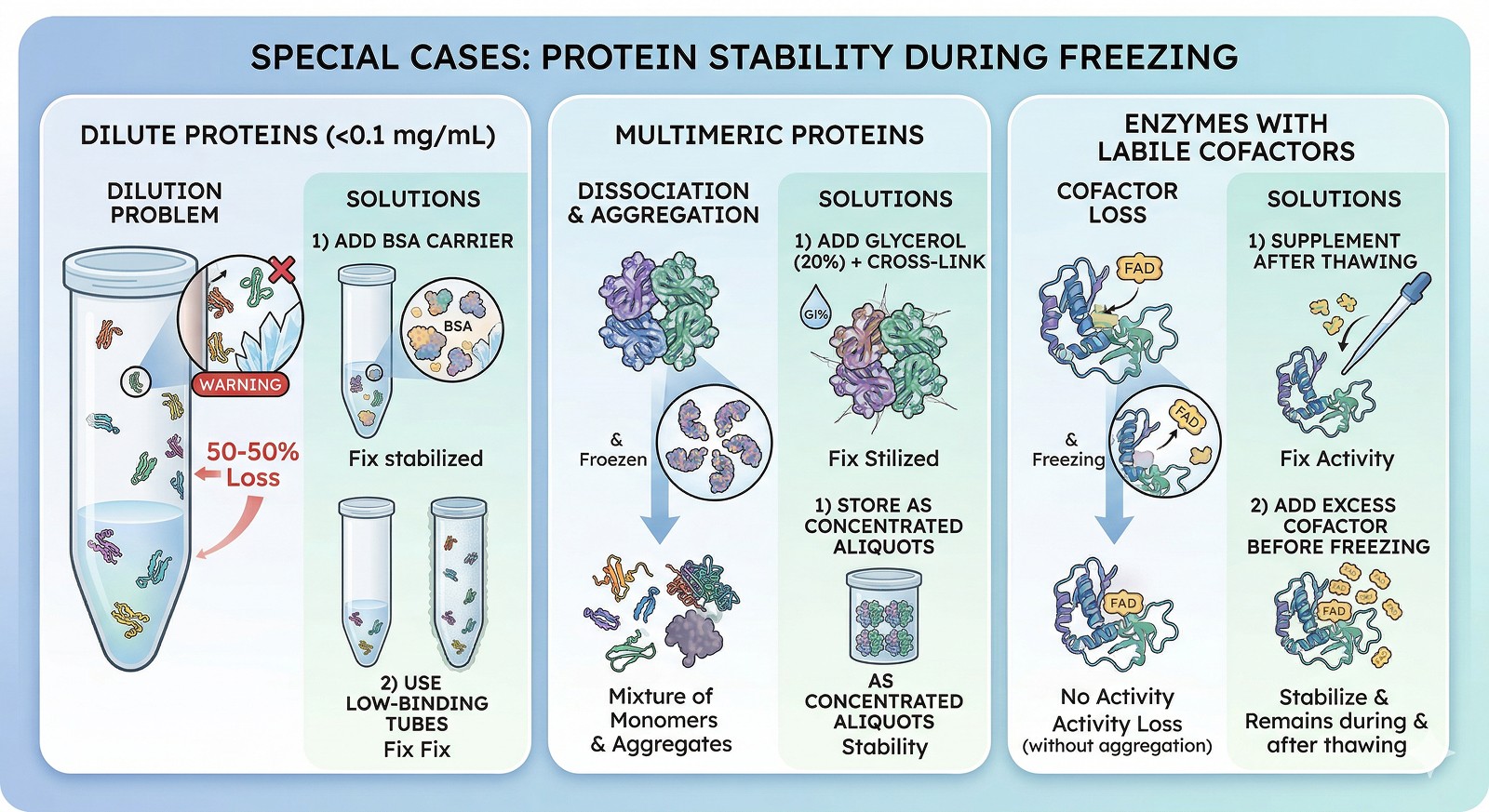
Dilute Proteins (<0.1 mg/mL)
At low concentrations, surface effects dominate:
Protein adsorbs to tube walls and ice surfaces
Loss can be 50–90% from a single freeze-thaw
Fix: Add 0.1–1 mg/mL BSA as a carrier. Or use low-binding tubes (Eppendorf Protein LoBind).
Multimeric Proteins
Oligomeric proteins can dissociate during freezing:
Freeze-concentration disrupts weak subunit interfaces
Thawed protein may be a mixture of monomers and aggregates
Fix: Add glycerol (20%) + cross-link if the complex is critical. Or store as concentrated aliquots where the high protein concentration drives reassembly.
Enzymes with Labile Cofactors
Some cofactors dissociate during freezing:
PLP (pyridoxal phosphate), FAD, FMN, metal ions
Activity loss without apparent aggregation or denaturation
Fix: Supplement with cofactor after thawing. Add excess cofactor before freezing.
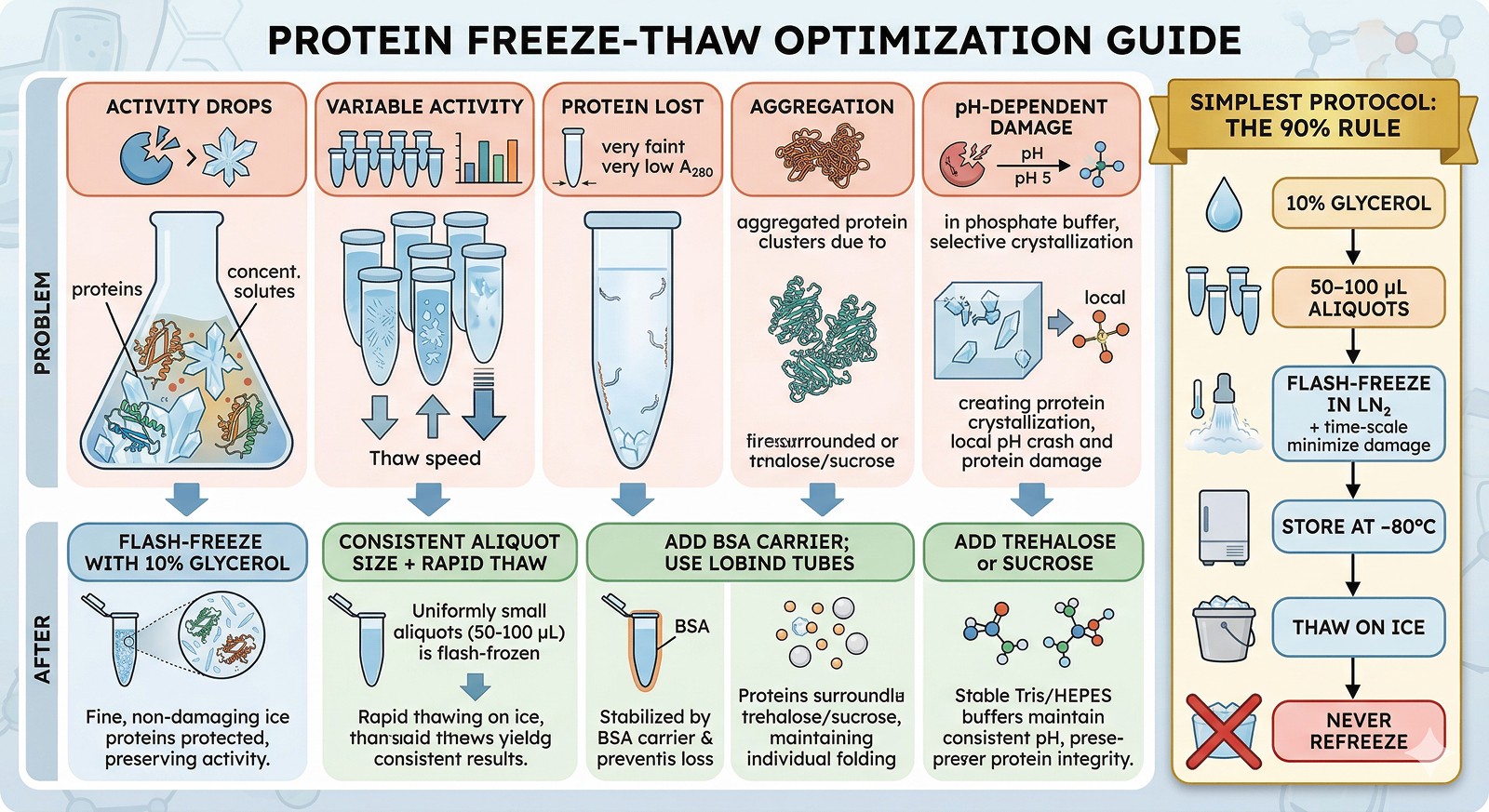
The Bottom Line
Problem | Cause | Prevention |
|---|---|---|
Activity drops after freeze-thaw | Ice crystal damage + freeze-concentration | Flash-freeze with 10% glycerol in single-use aliquots |
Variable activity between aliquots | Uneven freezing, different thaw speeds | Consistent aliquot size + flash-freeze + rapid thaw |
Protein lost (low A280 after thaw) | Surface adsorption at low concentration | Add BSA carrier; use LoBind tubes |
Aggregation after thawing | Freeze-concentration-induced aggregation | Add trehalose or sucrose; lower protein concentration |
pH-dependent damage | Phosphate buffer pH crash | Switch to Tris or HEPES |
The simplest protocol that works for 90% of proteins: 10% glycerol, 50–100 µL aliquots, flash-freeze in LN₂, store at –80°C, thaw on ice, never refreeze.
Stability-Aware Protein Handling
Orbion's Bench module generates formulation and storage protocols that account for predicted protein properties—aggregation propensity from AstraUNFOLD, thermal stability estimates, and buffer compatibility. When you know your protein's specific vulnerabilities before purification, you can design a storage strategy that preserves activity from day one.
References
Chang BS, et al. (2009). Mechanism of protein damage during freezing and dehydration and its prevention. Journal of Pharmaceutical Sciences, 98(9):2886-2908. Link
Pikal-Cleland KA, et al. (2000). Protein denaturation during freezing and thawing in phosphate buffer systems: monomeric and tetrameric β-galactosidase. International Journal of Pharmaceutics, 196(1):65-78. Link
Bhatnagar BS, et al. (2007). Protein stability during freezing: separation of stresses and mechanisms of protein stabilization. Pharmaceutical Development and Technology, 12(5):505-523. Link
Carpenter JF, et al. (1997). Rational design of stable lyophilized protein formulations: some practical advice. Pharmaceutical Research, 14:969-975. Link
